4 promising heart failure therapies interventional cardiologists should keep an eye on
Medical researchers have stayed busy in recent years working to develop new interventional heart failure treatments that offer more relief than guideline-directed medical therapy (GDMT) without requiring a left ventricular assist device (LVAD) or heart transplant. The interventional cardiologists at Cleveland Clinic’s Sydell and Arnold Miller Family Heart, Vascular and Thoracic Institute, for instance, are involved in multiple clinical trials exploring these promising therapies.
Cleveland Clinic, regularly voted the No. 1 heart hospital in the United States, provided an update on some of these trials, noting that “the treatment landscape between GDMT and LVAD placement or heart transplant can be broad and challenging.”
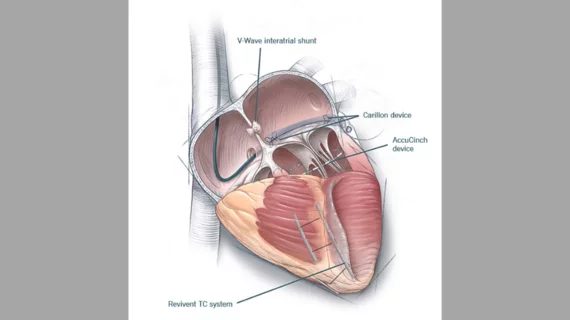
1. The Carillon Mitral Contour System (Cardiac Dimensions)
The EMPOWER trial is focused on the Carillon Mitral Contour System from Cardiac Dimensions, which has already gained CE mark approval in Europe for treating functional mitral regurgitation (MR). This device is place in the right heart using transcatheter access through the jugular vein; it was designed to improve heart function by reshaping the patient’s mitral valve annulus and reducing mitral annular dilation.
Samir Kapadia, MD, an interventional cardiologist and Cleveland Clinic’s chair of cardiovascular medicine, is helping lead the EMPOWER trial. He and his team hope to enroll a total of 300 patients who have heart failure with mild functional MR. Patients will be randomized to receive either the Carillon device or a sham device, and they will be followed for a planned total of five years.
“This is an extremely large patient population that is not currently being addressed by studies of other novel therapies,” Kapadia said.
2. The AccuCinch Ventricular Restoration System (Ancora Heart)
The CORCINCH-HF study is focused on Ancora Heart’s AccuCinch Ventricular Restoration System, an interventional device that is implanted using transfemoral access and “clinches” the upper part of the patient’s heart. Implantation follows a workflow comparable to transcatheter mitral annuloplasty procedures.
Rishi Puri, MD, PhD, an interventional cardiologist with Cleveland Clinic, is taking part in the study, which is designed to include 400 patients from up to 80 different facilities. Outcomes from the AccuCinch and GDMT together will be compared with outcomes from just GDMT. Patients will be followed for at least two years.
“The aim is to reduce left ventricular wall stress and dimensions, initiating a biologic process of reverse remodeling to improve myocardial contractility and overall function,” Puri said. “This should improve the patient’s quality of life and survival, increase exercise capacity and reduce heart failure hospitalizations.”
3. The V-Wave Ventura Interatrial Shunt System (V-Wave)
The RELIEVE-HF study is focused on the V-Wave Ventura Interatrial Shunt System from V-Wave. The device was designed to reduce left-sided cardiac filling pressure in patients with advanced heart failure by shunting blood across the interatrial septum from the left atrium to the right atrium. Providers place the device using fluoroscopic and echocardiographic guidance.
One key detail about this device’s design, according to Cleveland Clinic’s analysis, is the smaller shunt size.
“This is important because a previous interatrial shunt, which was larger, did not perform well in clinical trials, and this was believed to be due in part to its size,” Kapadia said.
The RELIEF-HF study aims to enroll 500 patients to either undergo the procedure and GDMT or GDMT alone. All patients will undergo diagnostic right heart catheterization and invasive echocardiography to “enable masking.” Shunt recipients will be followed for a total of five years.
4. The Revivent TC Transcatheter Ventricular Enhancement (BioVentrix)
The ALIVE trial is focused on the Revivent TC Transcatheter Ventricular Enhancement from BioVentrix. The device was developed to be an interventional treatment option for ischemic cardiomyopathy with left ventricular scarring, potentially giving clinicians a way to help these patients without an advanced surgical procedure. Two sets of micro-anchors are placed in the patient’s scarred heart: internal micro-anchors are placed into the interventricular septum by an interventional cardiologist and external micro-anchors are placed in the left ventricle’s outer wall by a cardiac surgeon. When these pairs are drawn to one another, it helps the left ventricular wall take on “a more normal shape and size.”
Edward Soltesz, MD, the surgical director of Cleveland Clinic’s Kaufman Center for Heart Failure Treatment and Recovery, said he hopes this treatment can “slow cardiomyopathy progression and improve quality of life.” The ALIVE trial will enroll 126 patients, which each patient expected to be followed for five years.
“The Revivent TC procedure offers a minimally invasive option to select patients with symptomatic ischemic heart failure without obviating advanced heart failure treatment options in the future,” added Jerry Estep, MD, chair of cardiovascular medicine at Cleveland Clinic Florida and the trial’s co-principal investigator. “If the trial results are positive, it will provide an appealing solution to the scar tissue that is the root cause of left ventricular dysfunction and disease progression in these patients.”
Read the full Cleveland Clinic ConsultQD update here.
Related Interventional Cardiology Content:
VIDEO: HeartFlow FFR-CT sees increased interest after inclusion in the 2021 Chest Pain Guidelines
4 support catheters receive FDA clearance for coronary use
Q&A: New ACC President Edward Fry discusses the past, present and future of cardiology
Interventional cardiologists complete first heart procedure of its kind in North America
Drug-coated balloons comparable to drug-eluting stents when PCI patients have a high bleeding risk